He threw himself into the water and swam out in search of the swans, who caught sight of him and hurried towards him, their feathers ruffled. "Kill me" cried the poor animal, hanging his head towards the surface of the water, awaiting death. But what did he see in the transparent water? He saw his own image beneath him, no longer that of an ugly, dirty grey duckling, but that of a majestic swan. There's no harm in being born in a farmyard when you hatch from a swan's egg.
Hans Christian Andersen, The Ugly Duckling
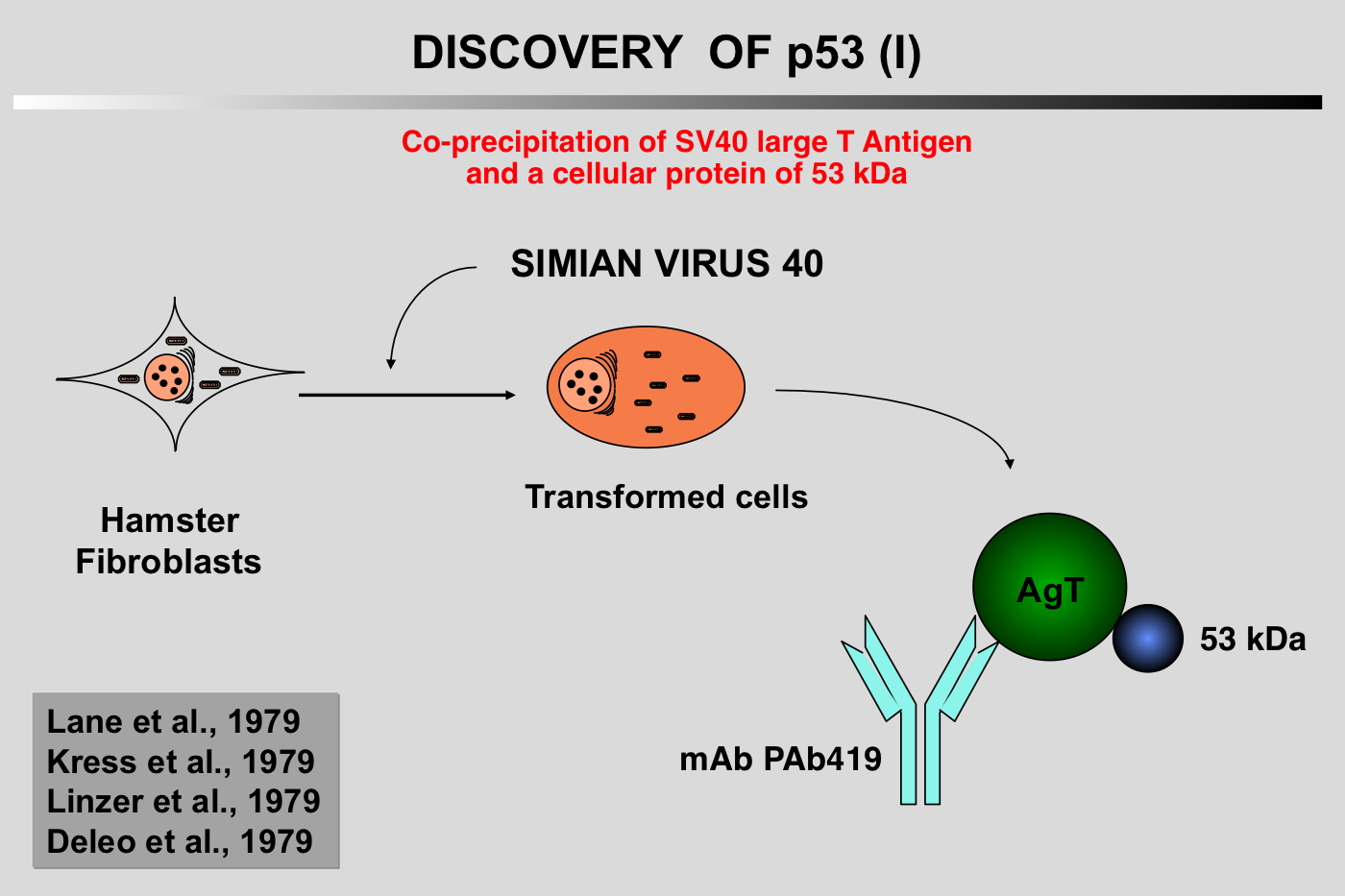
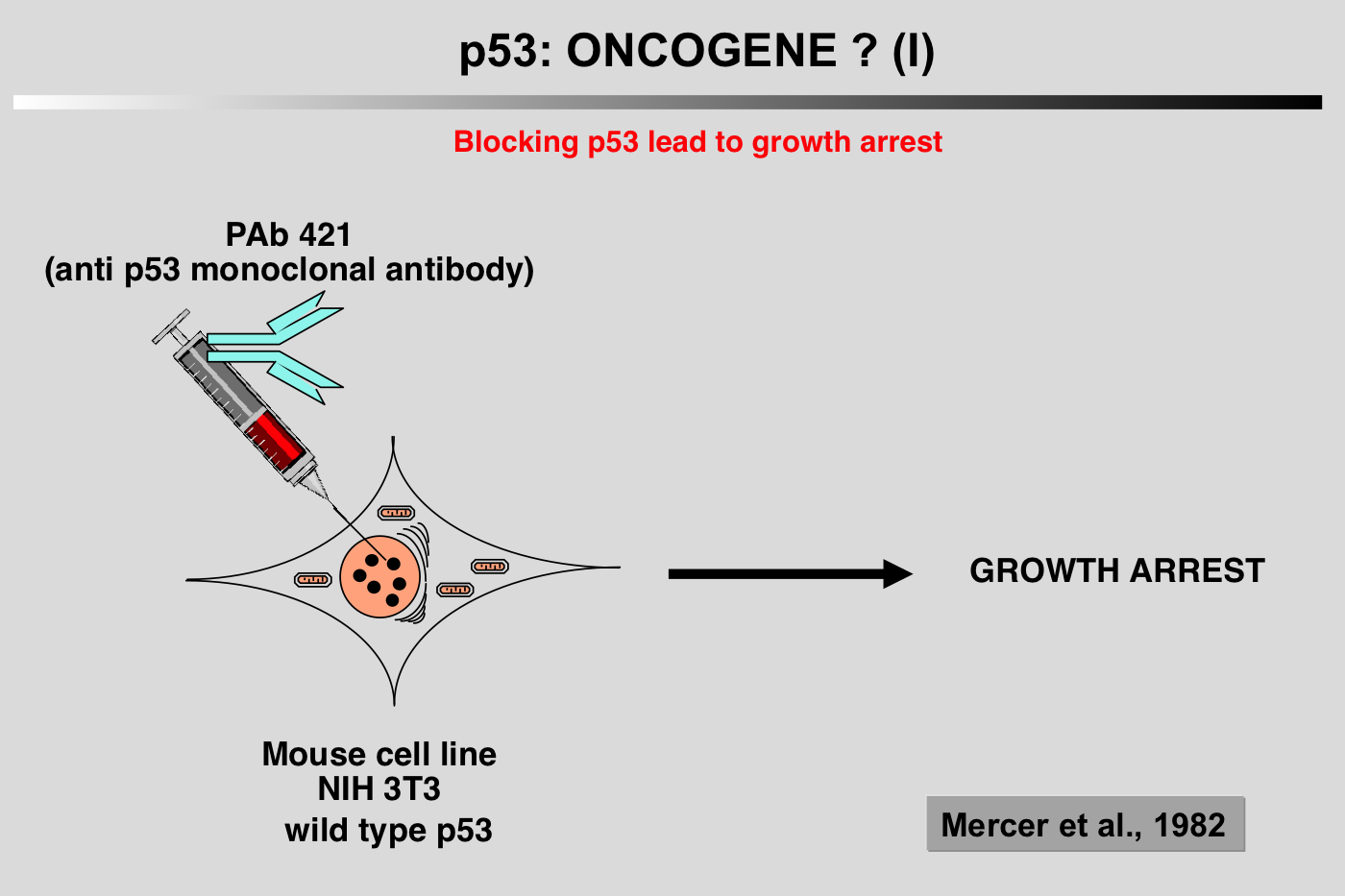
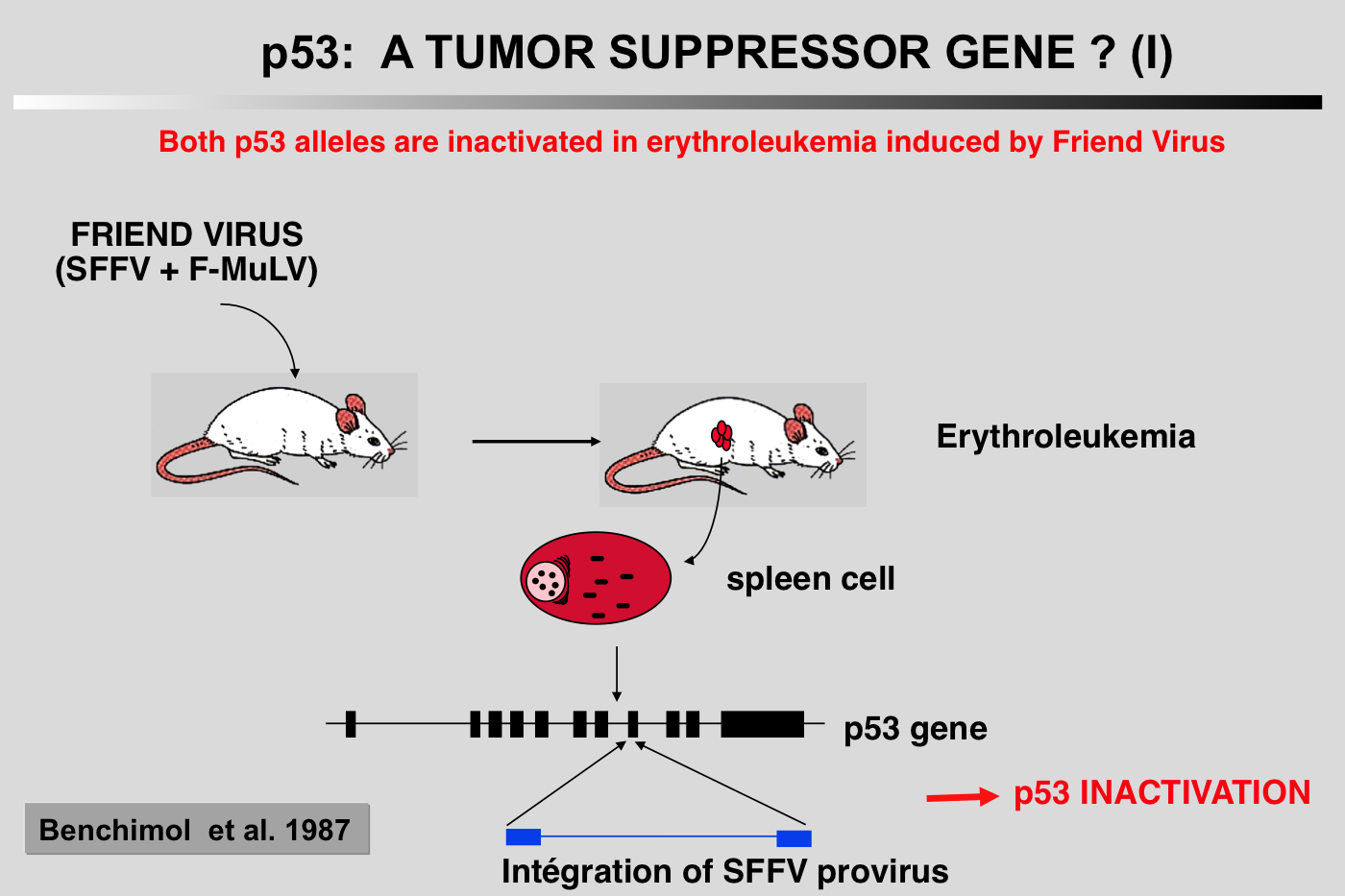
The history of p53 is a chaotic voyage from the world of oncogenes to the world of tumor suppressor genes, while retaining a certain degree of individuality. Apart from artifactual problems related to involuntary cloning of mutant p53, this ambiguity is also due to our propensity to over-categorize in order to satisfy our Cartesian and oversimplistic view of science.
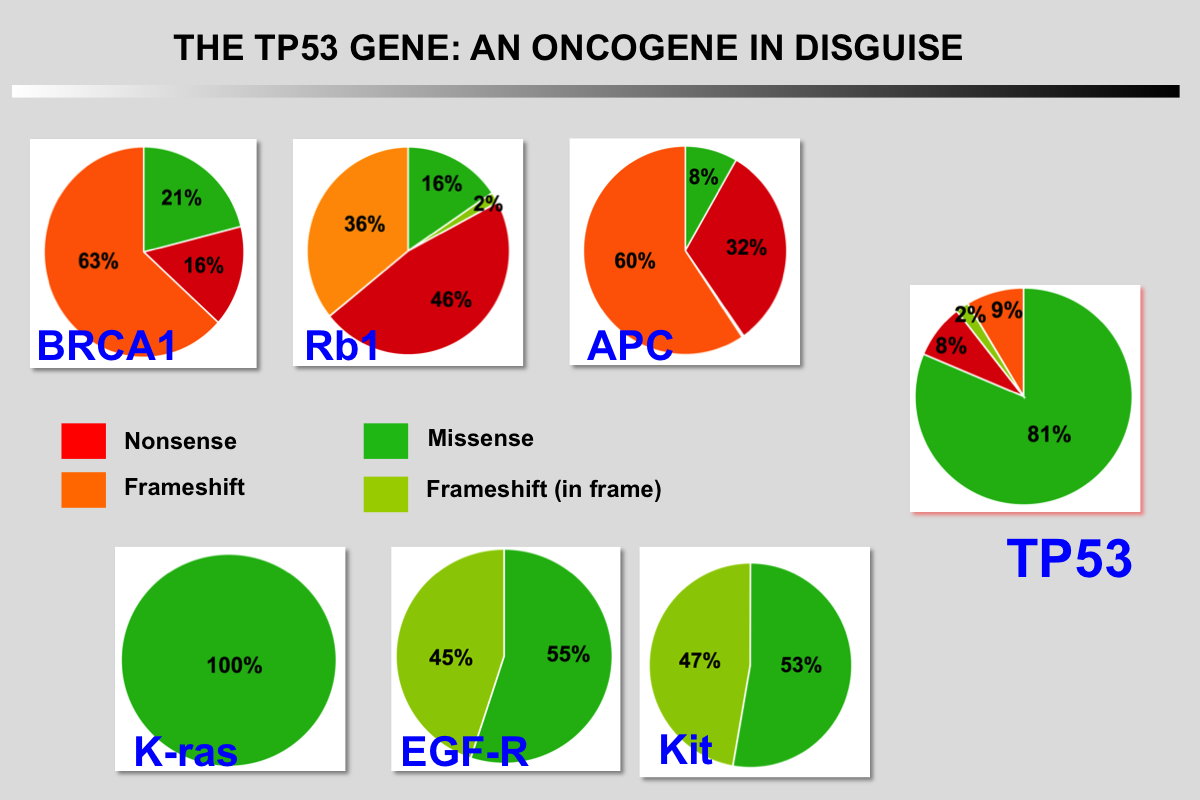
The idea that some p53 mutations can actively participate in cellular transformation was already postulated in 1990 and several arguments are in favor of such a model (Eliyahu et al., 1990; Lane and Benchimol, 1990); first of all, the mode of "inactivation" of wild-type p53. Unlike most other tumor suppressor genes that are inactivated by frameshift or nonsense mutations leading to disappearance or aberrant synthesis of the gene product, almost 90% of p53 gene mutations are missense mutations leading to the synthesis of a stable protein, lacking its specific DNA binding function and accumulating in the nucleus of tumor cells (Soussi and Béroud, 2001). This particular selection for accumulation of p53 mutations in tumor cells can have two consequences: i) a dominant negative role by hetero-oligomerization with wild-type p53 expressed by the second allele, or ii) a specific gain of function of mutant p53. Many studies have tried to distinguish between these two hypotheses, with no clear-cut conclusions (Michalovitz et al., 1991; Milner, 1995). This task is further complicated by the fact that not all p53 mutations appear to be equivalent and they present a marked heterogeneity of structure or loss of function. Transfection of various p53 mutations into cells devoid of endogenous p53 leads to an increase in their carcinogenicity, which varies according to the type of mutation (Dittmer et al., 1993; Halevy et al., 1990). This research into the oncogenic potential of certain p53 mutations is not purely theoretical, but has obvious clinical implications, as it could explain the marked disparity of the results of studies trying to demonstrate a relationship between the presence of a p53 gene mutation and various clinical parameters, such as survival or response to treatment (Muller and Vousden, 2013; Bieging et al., 2014; Khoo et al., 2014).
See below for more information.